3 results
Effects of a hard stop for C. difficile testing: Provider uptake and patient outcomes
- Danielle Doughman, David Weber, Nikolaos Mavrogiorgos, Shelley Summerlin-Long, Michael Swartwood, Alexander Commanday, Lisa Stancill, Nicholas Kane, Emily Sickbert-Bennett Vavalle
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, p. s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Clostridioides difficile infection (CDI) is a serious healthcare-associated infection responsible for >12,000 US deaths annually. Overtesting can lead to antibiotic overuse and potential patient harm when patients are colonized with C. difficile, but not infected, yet treated. National guidelines recommend when testing is appropriate; occasionally, guideline-noncompliant testing (GNCT) may be warranted. A multidisciplinary group at UNC Medical Center (UNCMC) including the antimicrobial stewardship program (ASP) used a best-practice alert in 2020 to improve diagnostic stewardship, to no effect. Evidence supports use of hard stops for this purpose, though less is known about provider acceptance. Methods: Beginning in May 2022, UNCMC implemented a hard stop in its electronic medical record system (EMR) for C. difficile GNCT orders, with exceptions to be approved by an ASP attending physician. Requests were retrospectively reviewed May–November 2022 to monitor for adverse patient outcomes and provider hard-stop compliance. The team exported data from the EMR (Epic Systems) and generated descriptive statistics in Microsoft Excel. Results: There were 85 GNCT orders during the study period. Most tests (62%) were reviewed by the ASP, and 38% sought non-ASP or no approval. Of the tests reviewed by the ASP, 33 (62%) were approved and 20 (38%) were not. Among tests not approved by the ASP, no patients subsequently received CDI-directed antibiotics, and 1 patient (5%) warranted same-admission CDI testing (negative). Of tests that circumvented ASP review, 18 (56%) ordering providers received a follow-up email from an associate chief medical officer to determine the rationale. No single response type dominated: 3 (17%) were unaware of the ASP review requirement, 2 (11%) indicated their patient’s uncharted refusal of laxatives, 2 (11%) indicated another patient-specific reason. Provider avoidance of the ASP approval mechanism decreased 38%, from 53% of noncompliant tests in month 1 to 33% of tests in month 6. Total tests orders dropped 15.5% from 1,129 during the same period in 2021 to 954 during the study period (95% CI, 13.4%–17.7%). Compliance with the guideline component requiring at least a 48-hour laxative-free interval prior to CDI testing increased from 85% (95% CI, 83%–87%) to 95% (95% CI, 93%–96%). CDI incidence rates decreased from 0.52 per 1,000 patient days (95% CI, 0.41–0.65) to 0.41 (95% CI, 0.32–0.53), though the change was neither significant at P = .05 nor attributable to any 1 intervention. Conclusions: Over time and with feedback to providers circumventing the exception process, providers accepted and used the hard stop, improving diagnostic stewardship and avoiding unneeded treatment.
Disclosures: None
Heterogeneous OPAT regimens within and across infection diagnoses: Day-level medication use patterns among 2072 OPAT patients
- Madison Ponder, Renae Boerneke, Asher Schranz, Michael Swartwood, Claire Farel, Alan Kinlaw
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s43-s44
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Patients receiving outpatient parenteral antimicrobial therapy (OPAT) are often medically complex and require carefully tailored treatments to address severe and often concomitant infections. Our objective was to illustrate the heterogeneity in antimicrobials used for patients in OPAT, within and across infection diagnosis groups. Methods: We abstracted electronic health record data regarding day-level treatment into a registry of 2,358 OPAT courses (n = 2,072 unique patients) treated in the University of North Carolina Medical Center OPAT program during 2015–2022 (total, 11,861 person weeks; average, 7 OPAT weeks per patient). We classified infection diagnoses into 10 hierarchical or mutually exclusive categories (eg, bacteremia only, diabetic foot infection (DFI) only, osteomyelitis only) (Fig., vertical axes). Accounting for 64 antimicrobial medications and 520 cocktails administered for at least 1 patient day in our OPAT registry, we also defined 18 hierarchical or mutually exclusive classifications of treatment (eg, “daptomycin alone” or “daptomycin and any other antibiotic(s)” (Fig. key). We conducted 2 stratified analyses to describe the heterogeneity across infection diagnoses with respect (1) to medications used at OPAT initiation (patient as unit of analysis) and (2) to medications used throughout OPAT (person time as unit of analysis, allowing for differential OPAT course to other treatment classifications during follow-up). We present stacked bar charts to visualize the intersection between infection diagnosis and treatment group. Results: Among patients in this OPAT registry, 34.6% had osteomyelitis and/or DFI, 4.8% had bacteremia, and 44.6% had multiple infections (Fig. 1). The most common medications in initial OPAT regimens were vancomycin (30.8% of OPAT patients), ceftriaxone (15.0%), and daptomycin (10.9%). We observed overall similarity between the distribution of treatment groups at initiation compared to cumulative person-time during the OPAT course (Figs. 1 and 2). However, we observed heterogeneity in medications by infection diagnosis (Figs. 1 and 2); for example, vancomycin was used in 39% of osteomyelitis cases but only 14% for endocarditis (Fig. 2). For several infection groups (eg, osteomyelitis, DFI, multiple infections, “other” single infections), no treatment classification exceeded 20% use (Figs. 1 and 2). Conclusions: Day-level data on medication use in this monitored registry of patients provided evidence of heterogeneity in the types of medications used throughout treatment in OPAT, which varies within and across infection diagnoses. These data highlight the need for multilayered ascertainment of medication exposure in this medically complex patient population to inform surveillance for adverse effects and guide comparative effectiveness research for postdischarge antibiotic treatment.
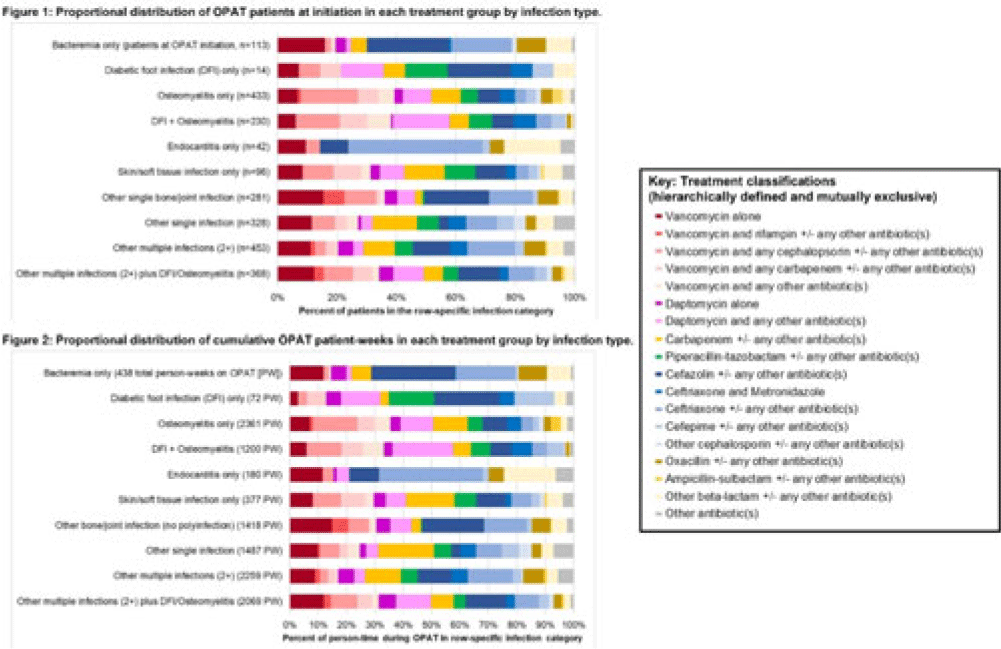
Disclosures: None
Implementation of Antibiotic Time Outs Using Quality Improvement Methodology
- Zachary Willis, May-Britt Sten, Lindsay Daniels, Jonathan Juliano, Michael Swartwood, Ronald Davis, Donna Krzastek, Clare Mock, Nikolaos Mavrogiorgos, Emily Sickbert-Bennett, David Jay Weber
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s275-s276
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Antibiotic time outs (ABTOs), formal reassessments of all new antimicrobial regimens by the care team, can optimize antimicrobial regimens, reducing antimicrobial overuse and potentially improving outcomes. Implementation of ABTOs is a substantial challenge. We used quality improvement methods to implement robust, meaningful, team-driven ABTOs in general medicine ward services. Methods: We identified and engaged stakeholders to serve as champions for the quality improvement initiative. On October 1, 2018, 2 internal medicine teaching services (services A and B), began conducting ABTOs on all patients admitted to their services receiving systemic antimicrobials for at least 36 hours. Eligible patients were usually identified by the team pharmacist. ABTOs were completed within 72 hours of antibiotic initiation and were documented in the electronic medical record (EMR) by providers using a template. The process was modified as necessary in response to feedback from frontline clinicians using plan-do-study-act (PDSA) methods. We subsequently spread the project to 2 additional internal medicine services (services C and D); 2 family medicine teams (services E and F); and 1 general pediatric service (service G). The project is ongoing. We collected data for the following metrics: (1) proportion of ABTO-eligible patients with an ABTO; (2) proportion of ABTOs conducted within the recommended time frame; (3) documented plan changes as a result of ABTO (eg, change IV antibiotics to PO); (4) proportion of documented plan changes actually completed within 24 hours. Results: Within 12 weeks, services A and B were successfully completing time outs in >80% of their patients. This target was consistently reached by services C, D, E, F, and G almost immediately following launch on those services. As of June 29, 2019, >80% of eligible patients across all participating services have had a time out conducted for 16 consecutive weeks. ABTOs have resulted in a change in management in 35% of cases, including IV-to-PO change in 19% of cases and discontinuation in 5%. Overall, 77% of time outs occurred during the 36–72-hour window. Ultimately, 95% of documented plan changes were completed within 24 hours. Conclusions: ABTOs are effective but implementation is challenging. We achieved high compliance with ABTOs without using electronic reminders. Our results suggest that ABTOs were impactful in the non–critical-care general medicine setting. Next steps include (1) development of EMR-based tools to facilitate identifying eligible patients and ABTO documentation; (2) continued spread through our health care system; and (3) analysis of ABTO impact using ABTO-unexposed patients as a control group.
Funding: None
Disclosures: None




